|
1/19/2024 0 Comments Mg group number periodic table 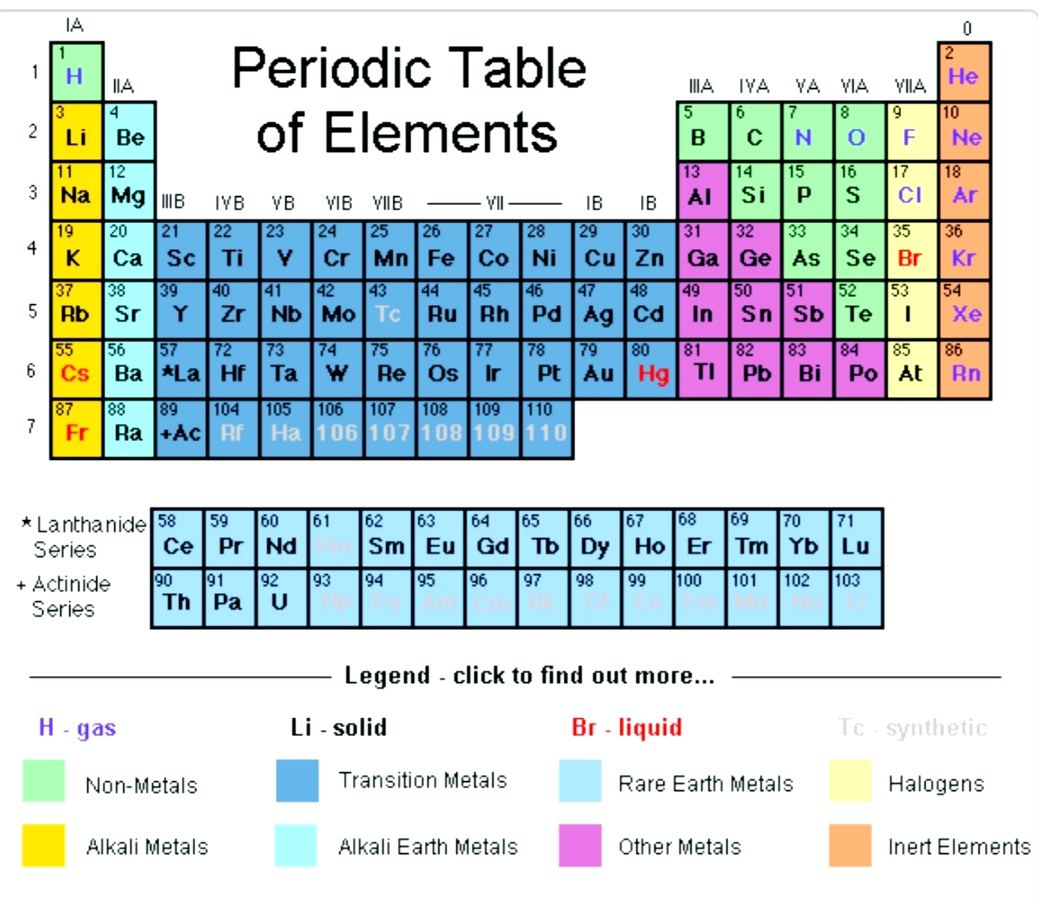
The electronic configuration of Magnesium will be 1s2 2s2 2p6 3s2. How do you write the electron configuration for Magnesium? What is the electronic configuration of Magnesium 12? What is the boiling Point of Magnesium in Kelvin?īoiling Point of Magnesium in Kelvin is 1363 K. Melting Point of Magnesium in Kelvin is 923 K. The alkaline earth metals include beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). What is the melting Point of Magnesium in Kelvin? What is the boiling Point of Magnesium?īoiling Point of Magnesium is 1363 K. Magnesium has 12 electrons out of which 2 valence electrons are present in the 3s2 outer orbitals of atom. How many valence electrons does a Magnesium atom have? The element Magnesium was discovered by J. It is located in group 2 and period 3 in the modern periodic table. Magnesium is the 12 element on the periodic table. It is essential for living organisms, and is used in a number of. We find Magnesium in the second group in the periodic table. Silvery metallic element belonging to group 2 of the periodic table (alkaline-earth metals). Magnesium is a chemical element with the symbol Mg and atomic number 12. Magnesium is a Group 2 chemical element with symbol Mg & atomic number 12. What is the position of Magnesium in the Periodic Table? Magnesium is a chemical element with symbol Mg and atomic number 12. Every hydrogen atom has one proton in its nucleus. Hydrogen, at the upper left of the table, has an atomic number of 1. In this table, an element's atomic number is indicated above the elemental symbol. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. The abbreviated electronic configuration of Magnesium is 3s2. What is the abbreviated electronic configuration of Magnesium? The electronic configuration of Magnesium is 1s2 2s2 2p6 3s2. 
For example, group 17 elements (one group left of the noble gases) form 1 ions group. Moving from the far right to the left on the periodic table, elements often form anions with a negative charge equal to the number of groups moved left from the noble gases. What is the electronic configuration of Magnesium? That is, group 1 elements form 1+ ions group 2 elements form 2+ ions, and so on. Magnesium Thermal Properties - Enthalpies and thermodynamics Optical Properties of Magnesium Refractive IndexĪcoustic Properties of Magnesium Speed of Sound Magnesium Magnetic Properties Magnetic Type Magnesium Heat and Conduction Properties Thermal Conductivity Refer to table below for the Electrical properties ofMagnesium Electrical Conductivity Hardness of Magnesium - Tests to Measure of Hardness of Element Mohs Hardness Refer to below table for Magnesium Physical Properties Densityġ.738 g/cm3(when liquid at m.p density is $1.584 g/cm3) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
